Ch2cl2 molecular geometry6/18/2023  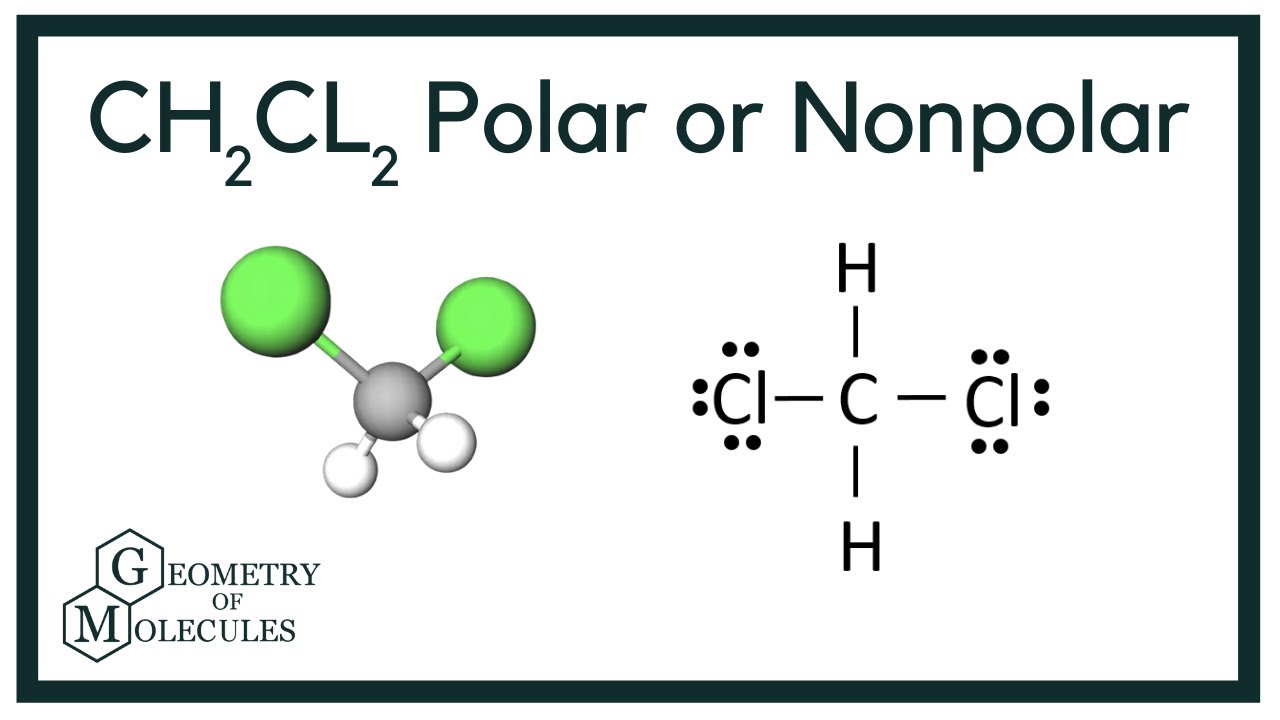
Octet rule states that an element is surrounded by eight electrobs in its Lewis structure.Īnalysing the Lewis structure of CH2Cl2 it is clear that it does not violate the octet rule. That’s because the molecule is actually tetrahedral in shape and not flat as is classically drawn in Lewis structure. Moving the chlorine atoms around does not produce a new structure. 
There is no other different acceptable lewis structure for CH2Cl2. Thus hydrogenĬan only fulfil duplet (having two electrons in its valence shell) rather than octet.Ĭarbon forms sigma bond ( single bond) with two hydrogenand chlorine atoms as shown in Lewis structure. 
Hydrogen has 1 electron in its 1s orbital and a s orbitalĬan only accommodate two electrons. Two hydrogen and two chlorine atoms share those 4 electrons with carbon to achieve the octet. Carbon needs 4 more electrons to complete its octet. 
In this CH 2Cl 2 molecule carbon is central atom( it has the highest bonding capacity and it is the shortest of the octet). The total number of valence shell electrons are 7 in chlorine so it needs 1 more electron to fulfill its octet. So 3s orbital will have two and 3p orital will have the last 5 electrons. Rest of the seven electrons will fill 3s and 3p orbitals respectively. 1s orbital contains 2, 2s orbital 2 and 2p contains 6 electrons. Hydrogen has atomic number 1, thus each H has 1 electron in its 1s orbital and needs 1 more to achieve duplet( to have 2 electrons in its valence shell) as 1s orbtal can only have two electrons. Thus carbon needs four more electrons to achieve its octet ( to have eight electrons in its valence shell that is outermost shell). The total numbers of its valence shell electrons of carbon is 4. Among these 6 electrons there are 2 electrons in its 1s orbital and 2 electrons in its 2s orbital and rest of the two electrons in its 2p orbital. Valence shell electrons of every atoms should be calculated first to determine the Lewis dot structure of any molecule.Ĭarbon has six electrons. Image credit: commons.wikimedia CH2Cl2 lewis structure valence electronsįrom the Lewis structure we can easily decide the number of bonds, lone pairs on each atom and be sure whether each of the atom fulfill their respective octer rule or not. The main agenda to determine the structure of any compound using this VSEPR theory that the atoms or substituents around the central atom will adopt such an arrangement that the repulsion between the valence shell electrons will be minimum.Īnalysing VSEPR theory it can be concluded that CH 2Cl 2 possesses a tetrahedral like molecular geometry (Methane like structure) as the carbon atom ( central atom) have two different types of atoms ( two hydrogen and two chlorine) around it. Molecular geometry of any molecule can be determined by using VSEPR (Valence Shell Electron Pair Repulsion) Theory. CH2Cl2 lewis structure molecular geometry In this article the lewis structure of CH2Cl2 and other detailed facts are discussed briefly. Melting point and boiling point of CH 2Cl 2 are -96.7 and 39.6 degree celcius respectively. We discuss the molecular geometry, hybridization, bond angle, octet rule, dipole moment and other related facts in detail and some frequently asked questions.ĭichloromethane ,mainly called as methylene chloride, is a polar organic solvent that is mostly used in various type of chemical research and manufacturing purpose also. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
